What are post translational modifications?
Post translational modifications are chemical changes to a protein that have an effect on its localization, function, and ability to interact with other molecules. These changes result in an extremely diversity among proteins in a cell and provide a means of regulating cellular activities. Modifications can be reversible or permanent. [1] Global screening of post translational modifications can reveal important residues and changes in a protein that results in a disease or condition.
There are several different kinds of post translational modifications. One important type of modification is called phosphorylation. Phosphorylation occurs when a phosphoryl group is added to a protein, usually at a serine, threonine, or tyrosine residue. This reversible process plays an important role in many cell functions, such as apoptosis, cell cycle regulation, and cellular growth, among others. [1][2]
Post translational modifications of FERMT1
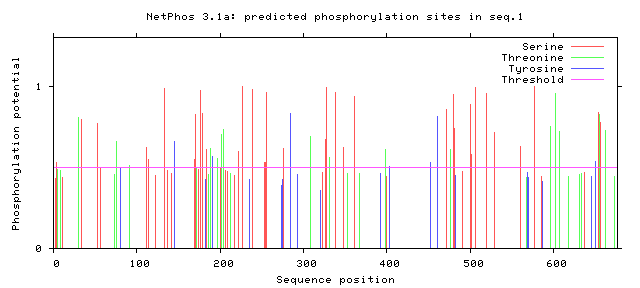
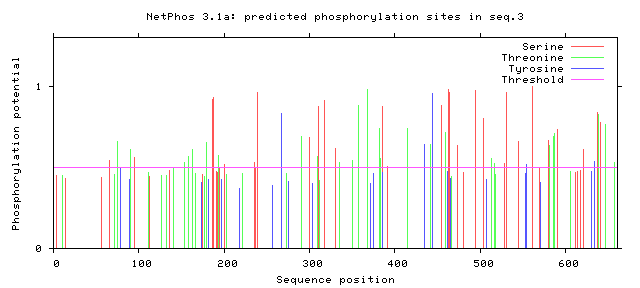
Using NetPhos, a database that can be used to predict phosphorylation sites in a protein sequence, the protein sequences of human FERMT1 and zebrafish fermt1 were analyzed. Peaks above the pink "Threshold" line are considered statistically significant.
HUMAN
Figure 1: Human FERMT1 phosphorylation sites.
Zebrafish
Figure 2: Zebrafish fermt1 phosphorylation sites.
Discussion
While the entire profile of phosphorylation appears fairly different between humans and zebrafish, there are some conserved regions and motifs. These tend to fall in the FERM domain of the protein, which is a highly conserved structural domain. Although there are relatively few phosphorylation sites within the kindlin-2-N domain, which is responsible for the protein's interaction with integrin, this could be advantageous in an study because it could focus the investigation to a few important sites. Further investigation into phosphorylation of FERMT1 could reveal more interesting functions of this protein that are disturbed in Kindler syndrome patients.
REFERENCES
Header image: www.promega.com/resources/guides/protein-analysis/protein-expression-methods/
Figure 1 & 2: www.cbs.dtu.dk/cgi-bin/webface2.fcgi?jobid=5EB6496E0000276769EDF07C&wait=20
1. “Overview of Post-Translational Modification: Thermo Fisher Scientific - US.” Thermo Fisher Scientific - US. Retrieved 5/9/2020 from www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-post-translational-modification.html.
2. Helmenstine, Anne Marie. “What Phosphorylation Is and How It Works.” (August 15th, 2019) ThoughtCo. Retrieved 5/9/2020 from www.thoughtco.com/phosphorylation-definition-4140732.
Figure 1 & 2: www.cbs.dtu.dk/cgi-bin/webface2.fcgi?jobid=5EB6496E0000276769EDF07C&wait=20
1. “Overview of Post-Translational Modification: Thermo Fisher Scientific - US.” Thermo Fisher Scientific - US. Retrieved 5/9/2020 from www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-post-translational-modification.html.
2. Helmenstine, Anne Marie. “What Phosphorylation Is and How It Works.” (August 15th, 2019) ThoughtCo. Retrieved 5/9/2020 from www.thoughtco.com/phosphorylation-definition-4140732.
This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison."